INTRODUCTION
Anti-CD19 chimeric antigen receptor (CAR) autologous T-cell therapy tisagenlecleucel (CTL019) has shown efficacy in pivotal trials and real-world data in relapsed/refractory pediatric B-cell precursor acute lymphoblastic leukemia (r/r B-ALL). However, 30-50% of patients relapse, because of CD19 loss in leukemic cells, or persistence/function reduction of CTL019. To improve relapse free survival, multitargeting CAR-T cells, such as bicistronic anti-CD22 and anti-CD19 or single sequential infusion are being developed. We report the safety and efficacy of an in-house manufactured tandem anti-CD19/22 dual CAR-T cells in pediatric, adolescents and young adults (AYA) diagnosed with r/r B-ALL.
MATERIAL AND METHODS
Eight batches of the tandem anti-CD19/22 dual CAR-T cells were manufactured under Good Manufacturing Practices (GMP) and authorized under a compassionate use program by the Spanish Agency for Medicines and Health (AEMPS). The starting material in all cases was patient´s non mobilized apheresis. After, CD4 + and CD8 + T cells were immune selected, activated with CD3/CD28 beads and transduced with a CD19/CD22 lentiviral vector (from Miltenyi Biotec®). Cell expansion was performed in a CliniMACS Prodigy® device with TexMACs® culture medium supplemented with IL-7 and IL-15. During manufacturing, process and quality controls were carried out and the finished product was infused fresh or cryopreserved.
RESULTS
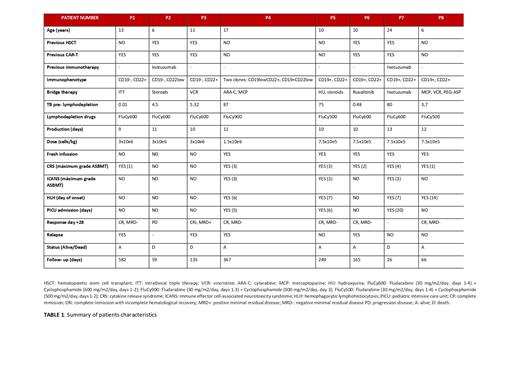
A total of 8 patients with a median age of 10.5 (range 6-25) years old received a median of 1 (range 1-3) doses of tandem anti-CD19/22 dual CAR-T cells containing a median of 1.13x106 (range 7.5x105-3x106) cells/kg infused. First 3 patients (37.5%) received cryopreserved product. Two patients (25%) were also diagnosed with Down syndrome (DS).
The median time between apheresis and infusion was 10.5 days (range 9-14 ). Five patients (62.5%) received previously tisagenlecleucel and four of them (50%) had also received a hematopoietic stem cell transplantation (HSCT). Leukemia blasts did not express CD19 in 2 patients (25%). Both received tisagenlecleucel previously, and one of them also expressed low levels of CD22. Median pre-infusion minimal residual disease (MRD) was 4.91% (range 0.01-87). Bridging therapy was low dose chemotherapy in 7 patients (87.5%), using inotuzumab in 1 patient. Lymphodepletion regimen was individualized, in 5 patients (62.5%) consisted on fludarabine 30 mg/m2/day for 4 days and cyclophosphamide 600 mg/m2/day for 2 days.
Regarding CAR cell product, mean transduction was 41.4 %. Similar CD4 +/CD8 + distribution was seen in most products generated (7/8, 88%), and central memory cells were the main subpopulation found, followed by naïve cells. Most CAR-T cell expansions (88%, median 7, range 7-28) were seen between day +7 and day +21 post-infusion.
Regarding toxicity, 7 patients (87.5%) presented cytokine release syndrome (CRS). Three patients (37.5 %) with high pre CAR-T tumor burden (TB) (range 75-87 %) developed CRS grade 3-4 (ASBMT guidelines) followed by ICANS grade 3.
On day +28 after infusion, 6 patients (75%) were in complete remission (CR), one of them with positive minimal residual disease (MRD). Two patients died because of disease progression (range 2-4.5 months) and a third one (with DS) because of toxicity before day +28 after CAR-T infusion. Consolidative HSCT was performed in 3 of 6 patients that achieved CR (50%) during the first 90 days after CAR-T cell infusion. During follow up of these transplanted patients (median 8.4 months, range 2.3-12.3), 1 patient (33.3%) relapsed after 11 months. One patient presented central nervous system relapse 1 month after CAR-T infusion and HSCT was performed and with 35 months of follow up, continues alive in CR. The last patient (with DS) relapsed 3 months after infusion and immunotherapy (inotuzumab) has been given in order to perform a second HSCT if CR is achieved.
CONCLUSIONS
In-house manufacturing of tandem anti-CD19/22 dual CAR-T cells is feasible and reproducible under GMP conditions in an academic institution.
Product infusion seems to be well tolerated in most patients, although patients with high TB developed high grade toxicities. Setting bridging therapy, reducing TB and manufacturing time remains a challenge.
Our experience suggests the efficacy of its use as a bridge to consolidative HSCT. Nevertheless, a larger number of patients in the context of a clinical trial would be necessary.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal